Table Of Contents:
Executive Summary
Inside the Chromatography Workflow
Lifecycle Value: One Platform, Multiple Workflows
Conclusion
References
Executive Summary
Biopharmaceutical process development faces two critical bottlenecks: the reliance on empirical, resource-heavy experiments, and the "Day One Reset" - where every new project requires significant planning because knowledge is trapped in silos. While mechanistic modeling offers a path to reduce experimental burden, it is often not scalable, typically requiring 4-6 weeks of setup by computational specialists for a single campaign.
ModelFlow transforms chromatography modeling from a bespoke task into a scalable Workflow. Our plug-and-play Chromatography Workflows allow teams to deploy physics-based simulations in minutes, not weeks. These Workflows provide:
Predefined Experimental Tasks: Guiding the bench team on exactly what data is needed.
Automated Data Pipelines: Ready-to-go connectivity and data pre-processing for unstructured Excel files, LIMS, databases, and ELNs.
Easier Insights with Web-Apps: Get a dedicated simulation tool packaged into a web-app for every project. This enables teams to get insights faster while reducing experimental burden, ensuring that process knowledge becomes a reusable asset.
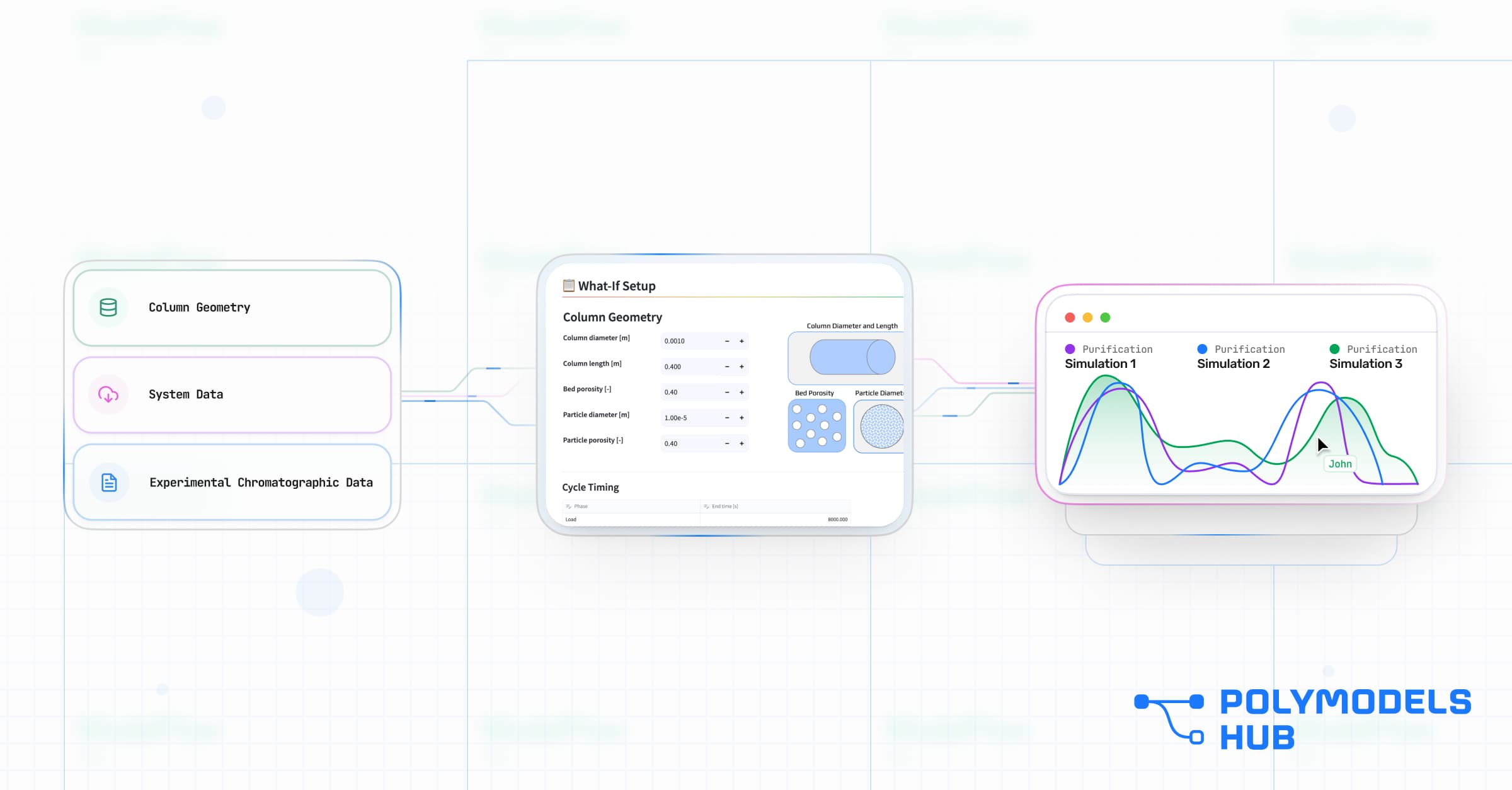
Inside the Chromatography Workflow
A workflow is more than just a simulation - it is a guided path from wet-lab to digital twin. By standardizing this path, ModelFlow allows you to move from data to decision in four clear steps.
Step 1: Predefined Experimental Tasks
The biggest barrier to adoption is uncertainty: "What data do I need to feed the model?" ModelFlow solves this by pre-populating the project with specific task lists.
Guided DoE / mb-DoE: The Workflow tells the user exactly which calibration runs are required - for example, specifying three specific flow rates to characterize mass transfer using state-of-art statistical and model-based design of experiment techniques.
Efficiency: This prevents "garbage in" and ensures the minimum number of wet-lab runs are used to achieve maximum predictive power removing any guesswork.
Step 2: Automated Data Pipelines
Manual data entry is the enemy of scalability. Our Workflows are designed to plug directly into your data ecosystem.
Connectivity: Pre-built connectors link ModelFlow directly to LIMS, ELNs, unstructured Excel files, internal databases, and other enterprise data sources. Your chromatography data connects seamlessly - without manual transfer or duplication.
Pre-processing & Data Cleaning: Laboratory data is rarely analysis-ready. It is fragmented across systems, inconsistently formatted, and often unvalidated. ModelFlow standardizes aggregation, validation, and cleansing through built-in data pre-processing tools - transforming raw experimental output into structured, model-ready datasets in just a few clicks.
Speed: Data flows directly from lab equipment and digital systems into the modeling pipeline. This eliminates transcription errors, reduces formatting time, and accelerates the path from experiment to insight.
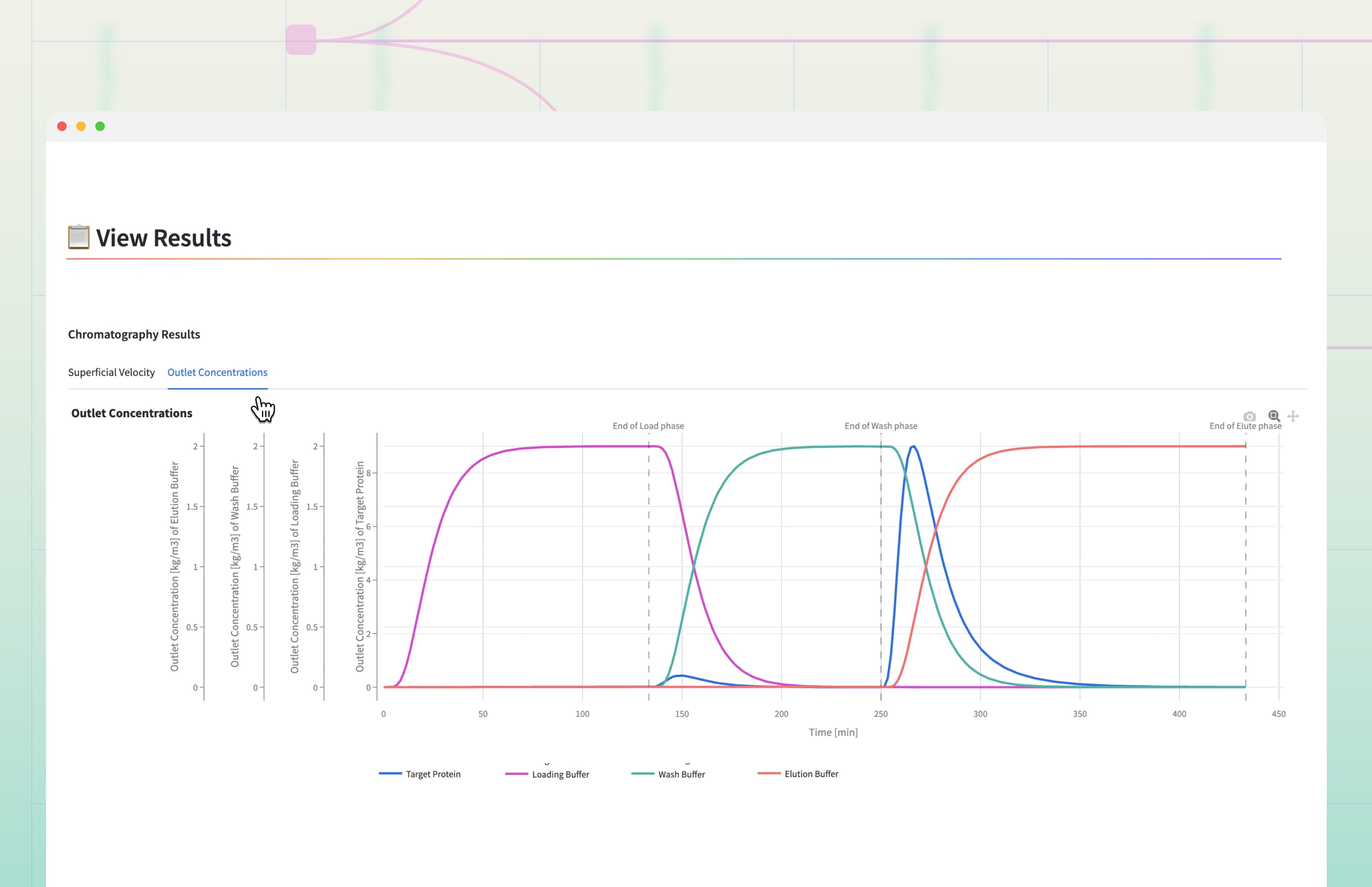
Step 3: A Physics-Based Toolkit for Every Separation
Beneath the intuitive Web-App lies a rigorous physics engine designed to handle the complexity of modern biopharmaceutical separations.
By modeling fundamental transport and binding phenomena (accumulation, convection, axial dispersion, mass transfer, adsorption, and desorption) ModelFlow creates a mechanistic foundation that is adaptable across all chromatography modes [1]-[6]. This framework allows you to precisely tailor simulations to your process by tuning the equilibrium relationship (the isotherm) to reflect the binding chemistry of your resin choice, the transport model to the column hydrodynamics, and the process schedule to your operating conditions.
This unified approach enables seamless deployment across:
Protein A / Affinity Chromatography - Target Capture
Anion Exchange Chromatography (AEX) - bind-and-elute or flow-through impurity clearance
Cation Exchange Chromatography (CEX) - charge variant separation and polishing
Flow-Through Impurity Removal Strategies
Buffer Tracking and Frontal Analysis
Because the physics remains consistent, knowledge becomes reusable. Once calibrated, the model evolves with your process - supporting resin changes, scale transitions, and operating window optimization without rebuilding from scratch.
Step 4: The Web-App Interface (Your Digital Lab Bench)
This is where ModelFlow fundamentally differs from traditional modeling tools.
Instead of requiring scientists to learn coding environments or install heavy desktop software and complex simulators, each Workflow comes with a user-friendly, scientist-oriented Web-App. The result is an intuitive simulation environment accessible across the organization through a standard web browser.
Scientists can run simulations, explore operating ranges, and evaluate process trade-offs without relying on specialist intervention. Modeling shifts from a bottleneck activity to an on-demand decision tool.
Customizable to Your Process
Each Web-App can be tailored to your specific Workflow, terminology, and decision needs. Whether you require predefined scenarios, constrained operating ranges, resin-specific configurations, or role-based access, the interface adapts to your organization - not the other way around.
By combining rigorous physics with accessible design, ModelFlow transforms simulation into a daily operational capability.
Lifecycle Value: One Platform, Multiple Workflows
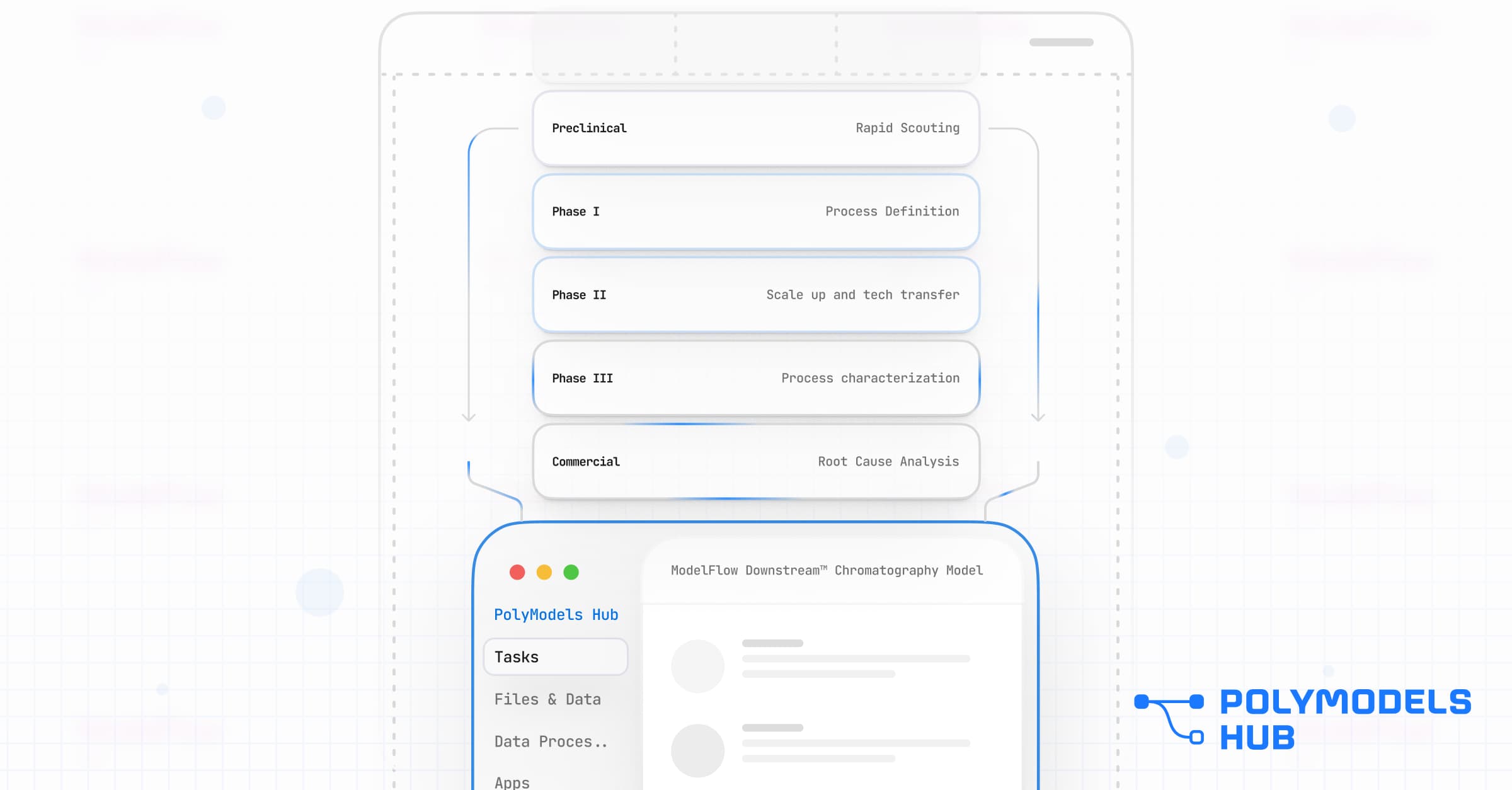
Because ModelFlow is modular, you can deploy different Workflows from the ModelVault for different stages of development, all powered by the same underlying physics. For example:
Preclinical: The Scouting Workflow
The Challenge: Multiple constructs may express well, but not all are actually purifiable at scale.
The Solution: A Scouting Workflow that uses mechanistic modeling to quantify downstream developability.
The Outcome: Identify the most robust candidate early, making data-backed decisions before committing resources to a molecule that is expensive to purify.
Phase I to III: The Tech Transfer Workflow
The Challenge: Scaling from the bench to the pilot plant carries high risk.
The Solution: A Scale-Up Workflow that acts as a Scalable Digital Twin.
The Outcome: Predict how changes in column diameter, bed height, or resin lot variability will impact Critical Quality Attributes (CQAs) before committing to expensive engineering runs.
Commercial: The Root Cause Workflow
The Challenge: A pressure spike or yield drop occurs in manufacturing.
The Solution: A Root Cause Analysis Workflow ready for rapid deployment.
The Outcome: Support data-driven release decisions. The model determines if the deviation impacts safety or efficacy, replacing qualitative risk assessments with physics-based evidence.
Conclusion
Chromatography is one of the most critical and knowledge-intensive steps in downstream development. It drives yield, purity, robustness, and ultimately cost of goods. Yet too often, its insights remain siloed within individual projects.
By turning chromatography into a scalable, physics-based Workflow, ModelFlow ensures that knowledge becomes reusable and expandable - not rebuilt from scratch each time.
Chromatography does not operate alone. Within the broader ModelFlow Downstream™ Library, chromatography Workflows integrate seamlessly with Tangential Flow Filtration and Viral Inactivation models. This allows teams to understand how performance in one unit operation impacts the next - connecting capture, polishing, and filtration into a coherent decision framework.
You can start with a single CEX or AEX step. Then expand progressively toward a fully connected downstream digital twin.
Not isolated simulations. But a structured, scalable foundation for downstream process intelligence.
References
Guiochon, G., Felinger, A., & Shirazi, D. G. G. (2006). Fundamentals of Preparative and Non-
linear Chromatography (2nd ed.). Academic PressFelinger, A., & Guiochon, G. (2004). Comparison of the Kinetic Models of Linear Chromatogra-
phy. Chromatographia, 60(Suppl 1), S175–S180Morbidelli, M., Servida, A., Storti, G., & Carra, S. (1982). Simulation of multicomponent ad-
sorption beds. Model analysis and numerical solution. Industrial & Engineering Chemistry Fun-
damentals, 21(2), 123-131Diedrich, S., Heymann, W., Lewek, S., Hunt, S., Tod, R., Kunert, C., Johnson, W., von Lieres,
E. (2017). Multi-state steric mass action model and case study on complex highloading behavior
of mAb on ion exchange tentacle resin. Journal of Chromatography A, 1525, 60-70Karlsson, D., Jakobsson, N., Axelsson, A., & Nilsson, B. (2004). Model-based optimization of a
preparative ion-exchange step for antibody purification. Journal of Chromatography A, 1055(1–2),
29–39Hitchcock, A. J., Rizzuto, R. S., & Crump, P. S. (2006). An improved kinetic Langmuir model
for ion exchange protein chromatography. Journal of Chromatography A, 1118(1–2), 149-156
